These ions and the atom of argon are known as isoelectronic. Notice that the three ions have electronic configurations identical to that of inert argon. The third lithium electron (this is the valence electron). The charges on the chlorine, potassium, and calcium ions result from a strong tendency of valence electrons to adopt the stable configuration of the inert gases, with completely filled electronic shells. electrons in the n 1 state, one with quantum numbers (1,0, 0, 1/2), the other with (1, 0, 0, 1/2).
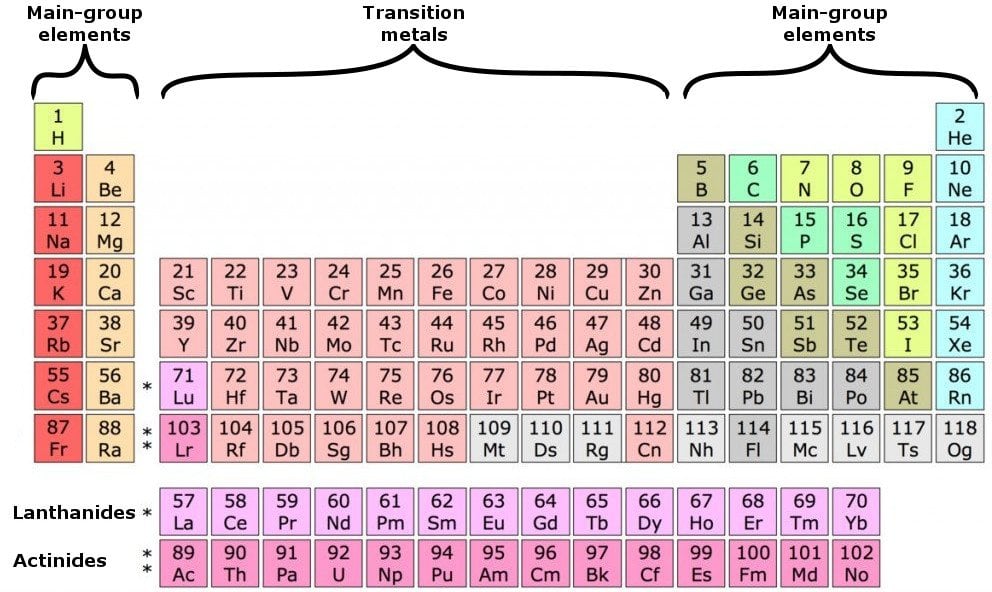

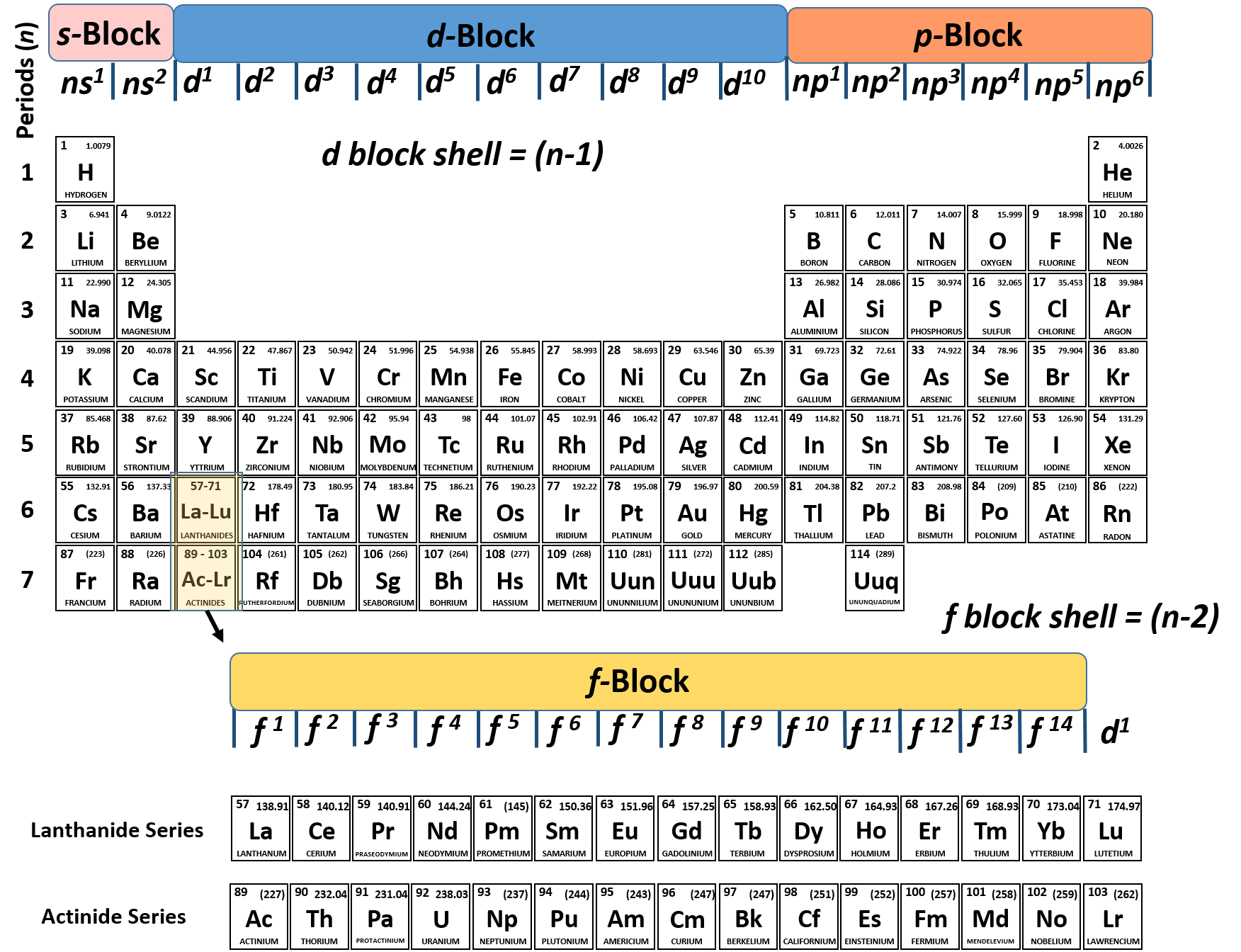
Table 2 compares three ions and a neutral atom. In Table, the common oxidation numbers in the last column are interpreted as the result of either losing the valence electrons (leaving a positive ion) or gaining enough electrons to fill that valence subshell. For example, in the H 2O molecule, each H has an oxidation number of +1, and the O is –2. In molecules, the various atoms are assigned chargelike values so the sum of the oxidation numbers equals the charge on the molecule. These are the valence electrons.įor ions, the valence equals the electrical charge. These electrons are most distant from the positive nucleus and, therefore, are most easily transferred between atoms in chemical reactions. For the transition metals with partially-filed d shells, valence electrons are those electrons outside the noble gas core. electrons of the d - block elements are considered to be valence electrons. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. Valence electrons are outer shell electrons for main group elements. periodic table, labeled 1-18 horizontally Groups 1, 2 s - subshell fills. The increasing positive charge attracts the electrons more strongly, pulling them closer to the nucleus. Valence electrons, which comprise the valence shell of the atom.įor brevity, many chemists record the electron configuration of an atom by giving only its outermost subshell, like 4 s 1 for potassium or 4 s 2 for calcium. In chemistry and physics, a valence electron is an electron associated with an atom that can form a chemical bond and participate in a chemical reactions. As we go across a period on the periodic table, however, electrons are being added to the same valence shell meanwhile, more protons are being added to the nucleus, so the positive charge of the nucleus is increasing. The electrons in the highest numbered subshells are the Study the third column of complete electronic configurations carefully so you understand how electrons are added to the subshell of lowest energy until it reaches its capacity then the subshell of the next energy level begins to be filled. The electronic configuration of an atom is given by listing its subshells with the number of electrons in each subshell, as shown in Tableġ. Quiz: Introduction to Oxidation-Reduction Reactions.Valence is the number of connections an atom tends to form. Introduction to Oxidation-Reduction Reactions Scientists soon observed patterns in the valence of the different elements.Quiz: Heat Capacities and Transformations.Quiz: Introduction to Organic Compounds.Quiz: Compounds with Additional Elements.
